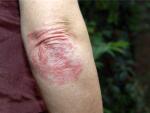
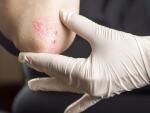
Your doctor may have explained why biologics were right for your psoriasis before you started treatment. But is there ever a reason to stop taking a biologic? When should you talk with your doctor about changing treatment?
Reasons You Might Stop
In most cases, once you start a biologic, it’s best not to stop, since most of these medications don’t work as well when restarted. Remicade (infliximab) is an example of a biologic that is typically used only once, since it often isn’t as effective when restarted after a break. Enbrel (etanercept) is an exception and seems better suited for off-and-on use.
However, like other psoriasis treatments, biologics can work for a while and then begin to lose effectiveness or cause unwanted side effects. It’s not unlikely that you may need to ask your doctor about switching treatments periodically.
Some of the main reasons you might stop taking a biologic include:
Failure to reach or to maintain an adequate response to the medication
Serious side effects
Pregnancy
Elective surgery
Vaccination
Getting Good Results
Getting a good response to biologic therapy varies widely—between 50 and 90 percent of patients experience positive results—and it varies by specific drug. The number of weeks before a significant positive response to a biologic occurs can range for each drug, as well.Several studies have shown that biologic therapies often work well for psoriasis patients during the first year of treatment.
Because of the gradual decreased response to biologics with time, your doctor may need to use a trial-and-error approach to find another biologic or combination of treatments that better controls your psoriasis.
Combination Therapy
Recent research has also shown that many people with psoriasis don’t get a good response with biologic treatment alone. Before you stop a biologic, your doctor may consider combining treatments to see if you get better results. Some research trials have shown that a combination approach with biologics and narrow-band ultraviolet B (NB-UVB) light therapy has led to a faster and better treatment response.
Studies of Enbrel (etanercept) with methotrexate have shown a much improved response compared to etanercept alone. Topical therapy and oral retinoids like Acitretin may be used in combination with biologics for improved results, too.
Pregnancy and Breastfeeding
Long-term animal studies with Enbrel (etanercept), Humira (adalimumab), and Remicade (infliximab) showed no evidence of harm to the fetus. Yet while the risk seems low, it’s still unknown what effect biologics may have on human pregnancy or on a nursing infant.
Because of the limited information, biologic drugs generally should be avoided in pregnancy. It’s generally thought that biologics should be avoided during breastfeeding, as well. However, there is limited evidence that Remicade (infliximab) is not excreted in breast milk.
It’s important to weigh the risks and benefits of taking biologics before, during, and after pregnancy with your doctors.
Prepping for Surgery
Whether and when you stop biologic therapy before elective surgery will depend on the specific drug that you’re on. It’s recommended that your biologic be stopped more than four half-lives before your surgery. A drug’s half-life is the time it takes for half the drug dose to be broken down into other substances in the body.
Each drug has a different half-life, so talk with your doctor to figure out when you need to stop your biologic before scheduling any elective surgery. Your biologic should be restarted after the procedure.
Vaccination Safety
People with psoriasis who are on biologic therapy should still receive the pneumococcal and annual influenza vaccines.
Recommendations for other vaccinations depend on whether it’s a live attenuated type or a killed inactive type. In general, no live vaccines should be given less than two weeks before and up to six months after starting biologics.
Inactivated vaccines are safe to receive while on biologics, but when possible, they should be given more than two weeks before starting treatment.
Before stopping or changing any of your psoriasis medications, it’s always important to talk with your doctor. Work with him or her to find the best treatment options to reduce or eliminate your symptoms.
Key Takeaways
It’s usually best not to stop taking a biologic, since most of these medications don’t work as well when restarted.
Reasons you might stop taking a biologic include failure to respond well to the medication, serious side effects, pregnancy, elective surgery, and necessary vaccinations.
Many people with psoriasis don’t get a good response with just biologic treatment alone. Your doctor may consider combining treatments for better results.